反义寡核苷酸对人淋巴瘤细胞系Namalwa细胞血管内皮生长因子表达影响的体外研究
作者:黎纬明,张敏,邹菁,童允洁,邹萍
【摘要】 为了研究硫代磷酸化修饰的血管内皮生长因子(vascular endothelial growth factor, VEGF)反义寡核苷酸(antisense oligodeoxynucleotide, ASODN)对人淋巴瘤细胞系Namalwa细胞VEGF表达的影响, 将终浓度分别为5、10、20 μmol/L的VEGF ASODN和错义序列与人淋巴瘤细胞系Namalwa细胞分别孵育24、48小时,采用RT?PCR检测VEGF mRNA的表达,采用链酶菌抗生素蛋白-过氧化酶免疫组织化学法(streptavidin/peroxidase, SP法)检测VEGF的表达。结果表明: VEGF ASODN 3个浓度组(5、10和20 μmol/L)处理的Namalwa细胞VEGF mRNA的表达分别为1.38、0.96、0.57, 错义序列组和对照组分别为1.79、1.84。当加入20 μmol/L VEGF ASODN作用48小时后,细胞内VEGF蛋白水平显著减少,而错义序列组Namalwa细胞VEGF蛋白水平未见明显改变。结论: VEGF ASODN在体外能够抑制Namalwa细胞VEGF的表达。
【关键词】 淋巴瘤
The recent studies have shown that in non?Hodgkin lymphoma patients with bone marrow involvement, the expression of vascular endothelial growth factor (VEGF) was associated with higher grading of NHL and high?grade transformation of low?grade lymphoma[1]. Data from Potti et al[2] indicated that signal transduction inhibition therapy using VEGF?inhibitors may have distinct effect for the patients with mantle cell lymphoma.
Bellamy et al[3] reported that VEGF over expressed in human lymphoma cell line Namalwa cells. We used VEGF ASODN and scrambled sequence transfecting Namalwa cell respectively, and confirmed the significant inhibition of VEGF ASODN on the VEGF mRNA expression, thus provided experimental and theoretical basis for the antisense gene therapy of lymphoma.
Materials and Methods
Drugs and reagents
VEGF ASODN sequence was designed based on the
human VEGF mRNA +261 to +281 nucleotides (5'? TGGCTTGAAGATGTACTCGAT?3'), and the scrambled (SCODN) sequence was 5'?TACGTAGTATGGTGTACGATC?3'[4]. All these were prepared in the suitable concentration with RPMI?1640 (purchased from Shanghai Biotechnology Ltd). The mouse anti?human VEGF monoclonal antibody was purchased from Santa Gruz Co. The SP kits were purchased from Beijing Zhongshan Biotechnology Ltd. The TRIZOL kits were purchased from Gibco, cDNA first?strand synthesis reaction kits were the products of MBI Co (purchased from Jingmei Biotech company). The dNTP and DNA enzyme were the products of Huamei Company.
Effect of Antisense Oligodeoxynucleotides on the Expression of Vascular Endothelial Growth Factor in Namalwa cell Cell culture and treatment of ASODN
Human lymphoma cell line Namalwa cells were obtained from Shanghai Biochemistry Cell Institute. Namalwa cells were cultured in RPMI?1640 (purchased from Gibco company) supplemented with 10﹪ fetal bovine serum ( products of Hangzhou Sijiqing Biological Engineering Material Co.),100 U/ml penicillin, 100 μg/ml streptomycin. They were cultivated in incubator of 37℃,5% CO2 and saturated humidity. Cells in log phase were inoculated in 6?well plates. Each plate consisting of 2 ml (cell density was 3×105/ ml) was added with VEGF ASODN and scramble sequence, whose final concentration were 5, 10 and 20 μmol/L respectively. Blank were also designed, each concentration was repeated for 3 times, incubated for 24 or 48 hours in normal condition.
RT?PCR for VEGF mRNA
Cells of each group incubating for 24 hours were collected and washed with PBS for 2 times.
Abstraction of cells RNA All procedures were based on the instruction of TRIZOL RNA abstraction kits. 5×106 cells were got and add with 1 ml TRIZOL, RNA was abstracted after chloroform delamination , avantin precipitation, washing and drying with 70% alcohol, and was dissolved with sufficient non?RNAase water, then the purity and quality of RNA were assessed using extreme ultraviolet spectroscopy and agarose gel electrophoresis, OD260/OD280=1.9, the RNA concentration was 0.5 μg/μl.
RNA reverse transcription The reverse transcription reactions of RNA were based on the instruction of cDNA first?strand synthesis reaction kit (MBI). 1 μl total RNA, 1 μl random primer and 10 μl DEPC water were mixed at 70℃ for 5 minutes; 5×reaction buffer 4 μl, RNase inhibitor 1 μl, 10 mmol/L dNTP 2 μl were added, mixed and warm bathed at 25℃ for 5 minutes; 1 μl M?Mulv reverse transcriptase was added and warm bathed at 25℃ for 10 minutes, 42℃ for 60 minutes, 70℃ for 10 minutes.
PCR reaction To use sufficient cDNA for PCR reaction. The primer sequence of VEGF[5]: sense: 5′?TCGGGCCTCCGAAACCATGA?3′, anti?sense: 5′?C?CTGGTGAGAGATCTGGTTC?3′; The primer sequence of β?MG: sense: 5′?ATCTTCAAACCTCCATGATG?3′, anti?sense: 5′?ACCCCCACTGAAAAAGATGA?3′. The reaction condition: 94℃ force?degeneration for 3 minutes; 94℃ degeneration for 1 minute, 55℃ renaturation for 50 seconds, 72℃ elongation for 40 seconds, these 3 steps circulating for 30 times, and then 72℃ elongation again for 5 minutes.
Relative quantitative analysis of PCR amplification products 10 μl PCR amplification products were detected through 1.5% agarose gel electrophoresis. The spot density scanning value of destination electrophoresis strip was obtained through FR200 image analysis software. The ratio of VEGF121 to β?MG was considered as the relative expression of VEGF. All relative amounts of anti?sense groups were compared with blank groups, the ratios were considered as parameters for the comparison of all groups. Analysis of interclass variance was used with SPSS 10.0.
SP immunohistochemistry for the expression of VEGF protein
Cells were cultured and manipulated as RT?PCR. Gather cells of each group after incubating for 48 hours, remove supernatant using centrifugation, re?suspense after PBS washing, adjust to proper density, and place Namalwa cells on glass slides (1 × polylisine), observe in microscope, fix with cold acetone for 15 minutes (4℃). Replace PBS with VEGF monoclonal antibody as blank control. Incubate with 3% hydrogen peroxide for 10 minutes, wash with distilled water, soak with PBS for 5 minutes, drop normal goat serum fluid 50 μl to incubate for 15 minutes, then pour it, add 50 μl mouse?anti?human monoclonal antibody (1:50 dilution), stay overnight in 4℃, wash with PBS for 3 minutes, 3 times, add biotin labeling goat?anti?mouse IgG 50 μl, incubate for 10 minutes at room temperature, wash with PBS for 3 minutes, 3 times, add horseradish enzyme labeling antibiotin fluid, incubate for 10 minutes at room temperature, wash with PBS for 3 minutes, 3 times, DAB coloration ,wash with tap water, afterstain with hematoxylin, routine dehydrate, clear with dimethyl benzene, mouting with neutron?resin.
Positive standard of VEGF: Cell which cytoplasm or membrane stain with buffy was positive, otherwise was negative. Select 10 fields in light microscope randomly, count 100 cells in each field. Count the number of positive cells. The exponent formula of positive cells is : (number of positive cells/1000) ×100%.
Result
Effect of VEGF ASODN on VEGF mRNA expression in Namalwa cells
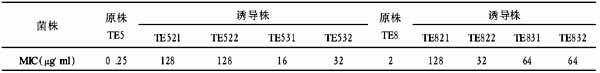
RT?PCR showed that the Namalwa cells expressed VEGF121 mRNA (516 bp) and VEGF165 mRNA(648 bp). The endogenous reference β?MG was 120 bp. The VEGF mRNA was measured by RT?PCR in cells treated with various concentrations of VEGF ASODN (5, 10, 20 μmol/L). After 24 hours, the band of VEGF165 mRNA 648 bp disappeared. With scanning the grey level ratio between VEGF 516 bp band and β?MG band, we detected the expression of VEGF121 mRNA treated with three concentration levels (5, 10, 20 μmol∕L) of ASODN were 1.38±0.21, 0.96±0.15, 0.57±0.09 respectively (F=12.38, p<0.05). ASODN showed a marked dose?dependent inhibition on the expression of VEGF. The expression of VEGF121 mRNA in PBS?treated cells and scrambled sequence treated cells were 1.79±0.28 and 1.84±0.31 respectively. There were no significant difference between these two groups (p>0.05). But difference was significant, as compared between the ASODN group and PBS?treated group (p<0.05)(Figure 1).
Figure 1. Expression of vascular endothelial growth factor mRNA in Namalwa cells treated by antisense oligodeoxynucleotides. M: marker. Lane 1,2: normal control. Lane 3,4: treated with scrambled sequence for 24 hours. Lane 5,6: treated with ASODN for 24 hours.
Expression of VEGF protein in Namalwa cells
The Namalwa cells were treated with VEGF ASODN and SCODN (20 μmol/L) for 48 hours. The PBS?treated group was used as blank control. The result showed that the VEGF expressed at high levels in SCODN treated group and control group, with the percentage of positive cells was 92.6% and 95.2% respectively. The cells treated with ASODN also expressed VEGF but at lower levels, with the percentage of 13.9%. Compared with the SCODN treated group and control group, the difference was statistically significant (p<0.05) (Figure 2). ASODN showed inhibitory activity, but SCODN had minimal inhibitory effect on the expression of VEGF in Namalwa cells, which in accordance with the result reported by RT?PCR, and further illustrated that the effect of ASODN was sequence?specific. Though the levels of VEGF expression were different in these three groups, the growth of Namalwa cells were roughly the same. The Namalwa cells were treated by ASODN (20 μmol/L), incubated for 24, 48 or 72 hours respectively, and the result showed the positive cells which expressed VEGF were 26.8﹪(24 hours), 13.9﹪(48 hours) and 19.4﹪(72 hours). The lowest expression of VEGF was occurred at 48 hours.
Figure 2. Expression of VEGF in VEGF ASODN or SODN treated cells (sp,×40). A: VEGF ASODN treated cells: VEGF was expressed in 13.9% cells. B: VEGF SODN treated cells: VEGF was expressed in 92.6% cells.
Discussion
As a key factor of tumor angiogenesis, VEGF can induce the endothelial cell proliferation and migration in vitro, and induce angiogenesis in vivo. VEGF was expressed in various solid tumor cells, and associated with prognosis [6]. To inhibit the expression of VEGF or block the pathway of its signal transduction can suppress the tumour angiogenesis[7]. Anti?VEGF antibody, soluble receptor or VEGF receptor tyrosine kinase inhibitor are now usually used to suppress the angiogenesis activity of VEGF[8]. But most of these drugs have short half?life periods in vivo, and their immune reaction also limited the repeatedly use. Antisense oligonucleotides blocking RNA to regulate the gene expression can also minimize the function of VEGF[9].
The VEGF ASODN sequence we used is complemented with the human mRNA of VEGF exon 3, the scrambled sequence was not homologous with the sequences in human genome. All these sequences were modified with phosprothioate to make them stable in blood serum. It took advantages as follows: (1) the target?specific inhibition on gene expression; (2)the possibility of full scale production, so it's more practical than antisense RNA mediated by vector. When Namalwa cells treated with the ASODN in vitro directly, it showed significant inhibition on the expression of VEGF mRNA and protein, and the inhibitory effect on the expression of VEGF mRNA was dose?dependent. PBS?treated cells and scrambled sequence treated cells had no effect. The results showed that the inhibitory effect was sequence?specific and dose?dependent. So VEGF ASODN might inhibit the angiogenesis of lymphoma, had inhibitory effect on the tumor growth, relapse and metastasis.
Our study showed that VEGF ASODN can suppress the expression of VEGF mRNA and protein in vitro, which may provide a theoretical basis on using VEGF ASODN as alone or combination with chemotherapy for the treatment of lymphoma.
【】
1Ho CL, Sheu LF, Li CY. Immunohistochemical expression of basic fibroblast growth factor,vascular endothelial growth factor, and their receptors in stage IV non?Hodgkin lymphoma. Appl Immunohistochem Mol Morphol, 2002; 10:316-321
2Potti A, Ganti AK, Kargas S, et al. Immunohistochemical detection of C?kit (CD117) and vascular endothelial growth factor (VEGF) overexpression in mantle cell lymphoma. Anticancer Res,2002; 22:2899-2901
3Bellamy WT, Richter L, Frutiger Y, et al. Expression of vascular endothelial growth factor and its receptors in hematopoietic malignancies. Cancer Res, 1999; 59:728-733
4Masood R, Cai J, Zheng T, et al. Vascular endothelial growth factor/vascular permeability factor is an autocrine growth factor for AIDS?Kaposi sarcoma. Proc Natl Acad Sci, 1997; 94:979-984
5Soker S, Gollamudi?Payne S, Fidder H, et al. Inhibition of vascular endothelial growth factor(VEGF)?induced endothelial cell proliferation by a peptide corresponding to the exon 7?encoded domain of VEGF165. J Biol Chem, 1997; 272:31582-31588
6Plate KH, Breier G, Weich HA, et al. Vascular endothelial growth factor is a potential tumour angiogenesis factor in human gliomas in vivo. Nature, 1992; 359(6398):845-848
7童允洁,张敏,邹萍等. VEGF硫代反义寡核苷酸抑制U937细胞VEGF的表达. 实验血液学杂志, 2004; 12 :151-153
8Gasparini G, Toi M, Gion M, et al. Prognostic significance of vascular endothelial growth factor protein in node?negative breast carcinoma. J Natl Cancer Inst, 1997; 89:139-147
9Nguyen JT, Wu P, Clouse ME, et al. Adeno?associated virus?mediated delivery of antiangiogenic factors as an antitumor strategy. Cancer Res, 1998; 58:5673-5677